- Lab Services
- Surface Roughness & 3D Topography
- Chemical
- Color Measurement
- Contact Angle
- Gloss Measurement
- Liquids
- Mechanical
- Abrasion
- Contact Stylus Surface Roughness Analysis
- Depth Profiling
- Express Property Mapping through Accelerated Nanoindentation
- Macroscratch
- Microindentation
- Microscratch
- Modulus Mapping
- NanoDMA
- Nanoindentation
- Nanoscratch
- Rheology
- Scratch Testing ISO 1518
- Shore A and Shore D Hardness
- Universal Testing Machine
- Metallurgy
- Microscopy
- Particle Size
- Peel Strength
- Pore Size
- Technical Consulting
- Thermal
- Tribological
- X-Ray Diffraction
- Zeta Potential
- Products
- Industries
- Resources
- About Us
- Testimonials
- Contact Us


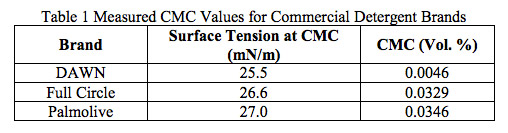
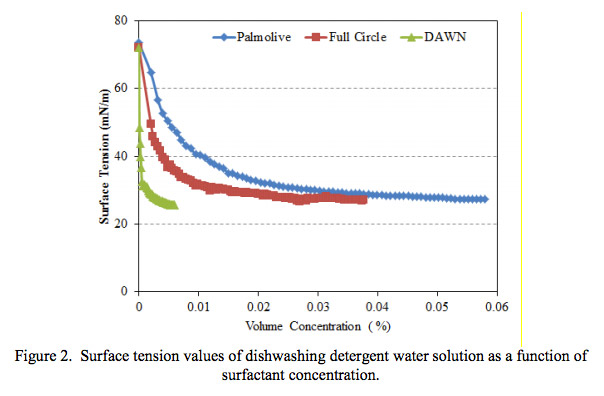
 Cosmetics and household products can be comprised of many different materials. It is crucial to know the material properties of each product in order to verify the functionality. At Ebatco, our expert scientists will provide accurate results in an efficient manner. You will receive a professional report with tables and graphs in order to provide all of the necessary knowledge to determine whether the product meets all of its desired specifications.
Cosmetics and household products can be comprised of many different materials. It is crucial to know the material properties of each product in order to verify the functionality. At Ebatco, our expert scientists will provide accurate results in an efficient manner. You will receive a professional report with tables and graphs in order to provide all of the necessary knowledge to determine whether the product meets all of its desired specifications.